Liquid scintillation spectrometry for radionuclide analysis in water and liquid samples
Liquid scintillation spectrometry is an effective method for radionuclide analysis in water and liquid samples, particularly for alpha- and beta-emitting radionuclides that are difficult to detect by gamma spectrometry.
Introduction
At the initial stage of studying environmental and technological samples with unknown radionuclide composition, a conventional approach is typically applied. This includes the determination of gross alpha and beta activity, as well as gamma spectrometric analysis.
However, measurements of gross alpha and beta activity do not provide information about the actual radionuclide composition of the sample. Gamma spectrometry using HPGe detectors is a fast and informative technique, but for a number of long-lived alpha- and beta-emitting radionuclides with high radiotoxicity - such as Sr-90, transuranium elements (TRE), and naturally occurring radionuclides including Ra, Th, U, Pb-210, and Po-210 - its capabilities are limited.
For this reason, further identification of such radionuclides is usually carried out using dedicated radiochemical separation methods, selected depending on the possible composition of the sample.
Liquid scintillation (LS) spectrometry, which offers several advantages compared to conventional radiometric techniques, provides an alternative approach for evaluating radionuclide composition in various types of samples. LS spectrometers are widely used for radionuclide activity measurements due to their high detection efficiency for alpha and beta radiation combined with low background levels.
The liquid scintillation counting (LSC) method allows estimation of the energy distribution of charged particles. At the same time, one of the key challenges in LS spectrometry is the correct decomposition of complex multicomponent instrumental spectra into their individual components.
The analysis of multicomponent radionuclide mixtures originating from environmental or technological systems is complicated by both the uncertainty of the sample composition and the continuous, overlapping nature of beta spectra.
Modern mathematical approaches for interpreting complex LS spectra, implemented in the software packages SpectraDec and ASW3L, make it possible to obtain rapid information about the composition of major alpha- and beta-emitting radionuclides. In many practical cases, it is sufficient to determine whether the activity of specific radionuclides exceeds predefined control or regulatory levels, which is a common task in radiation monitoring.
A liquid counting sample can be prepared either by direct mixing of a sample aliquot with a liquid scintillation cocktail, or by preliminary evaporation and concentration of the sample followed by mixing with the scintillator.
To improve sensitivity for tritium and radiocarbon screening, a combustion procedure using special furnaces (oxidizers) may also be applied. The resulting gases are captured in absorbents and subsequently mixed with the scintillation cocktail.
The described approach is implemented in the modern liquid scintillation spectrometer TRIEL, operating in combination with ASW3L and SpectraDec software. ASW3L is used for measurement control, data processing, and routine analytical tasks. In particular, the software enables centralized control of multiple TRIEL spectrometers combined into a unified measurement system, which is especially important for laboratories with high sample throughput.
SpectraDec is applied for more advanced spectral analysis tasks involving complex multicomponent mixtures.
The TRIEL spectrometer is compatible with a wide range of scintillation cocktails and operates over a broad range of detectable energies and quenching levels.
Measurements are performed in standard low-diffusion polyethylene vials with a volume of 20 mL.
The LS analysis method described here can be applied to a wide variety of sample types, including:
-
tap water
-
groundwater and borehole water
-
seawater
-
surface waters
-
mineral and drinking water
-
process water and wastewater
-
liquid waste and leakages
-
sludges and drilling fluids
Reference radionuclide solutions used for building the spectral library are supplied by certified European laboratories.
Radionuclide identification via modeling of complex liquid scintillation spectra
Liquid scintillation spectra are typically continuous and complex in shape, while their individual components associated with different radionuclides overlap over a wide energy range.
To interpret such spectra, the measured signal is represented as a combination of elementary spectra. This approach is based on modeling the initial spectra transformed into a grouped form through superposition of individual reference spectra taken from a previously created radionuclide library.
The model spectrum is constructed as a linear combination of normalized spectra corresponding to different radionuclides:

where:
Mᵢ is the model spectrum;
Mᵢ,ⱼ(T) is the library spectrum for radionuclide j, normalized to unit activity;
cⱼ is the contribution (fraction) of each component.
The coefficients cⱼ are determined in such a way that the model spectrum reproduces the measured sample spectrum with the highest possible accuracy.
Each spectrum depends on the quenching parameter T of the sample. The solution of the problem is obtained by minimizing a functional that describes the difference between the model spectrum and the measured spectrum. In contrast to traditional approaches, the entire spectrum is processed as a whole, without splitting into separate energy windows. This leads to improved sensitivity of the measurements.
Creation of the spectral library
The shape of the instrumental spectrum strongly depends on the properties of the prepared liquid counting sample.
A spectrum of a sample with higher quenching is shifted toward the low-energy region, often accompanied by significant deformation.
For the creation of a radionuclide library, direct measurements of spectra for each radionuclide are used. This library of reference spectra is formed for a specific spectrometer and given characteristics of counting samples by measuring standard radionuclide solutions at different quenching levels.

Spectra of radionuclides H-3, C-14, Sr-90+Y-90, Tc-99 and external standard Cs-137 from the calibration library.

Spectra of radionuclides Ni-63 C-14, Sr-90+Y-90, Am-241 and Pb-210 from the calibration library.
The analytical database (a library of spectra with different quenching levels) for each radionuclide is typically formed for 8–10 quenching levels using nitromethane (or another compound) as a chemical quenching agent.
For example, for the determination of natural radioactive series in water, the following sets of spectra were used:
U-238
Th-234 + Pa-234 (in equilibrium)
Th-232
Th-228
Pb-210
Bi-210
Po-210
U-235
Th-231 + Pa-231 (in equilibrium)
Ra-228 + Ac-228 (in equilibrium)
U-234
Th-230
Ra-226
Ra-226 + Rn-222 + Po-218 + Pb-214 + Bi-214 + Po-214 (in equilibrium)
Ra-224 + Rn-220 + Po-216 + Pb-212 + Bi-212 + Po-212 + Tl-208 (in equilibrium)
During processing of the measured spectrum, the shapes of individual library spectra are adjusted to the required quenching level by interpolation between two adjacent reference spectra.
At present, the analytical database (spectral library with quenching taken into account) includes more than 30 beta- and alpha-emitting radionuclides (both individual radionuclides and those in equilibrium with their progeny), which makes it possible to determine various radionuclides also after radiochemical separation. At the same time, it is possible to investigate samples with complex radionuclide composition, such as wastes (solid and liquid radioactive waste), waters from monitoring wells, and similar samples, using a screening approach after simple sample preparation procedures.
Calculation of radionuclide activity
In the course of liquid scintillation analysis, the activity of a radionuclide Aⱼ in the measured sample is determined through the parameter cⱼ, which characterizes the contribution of the given radionuclide to the spectrum.
Thus, a model spectrum is constructed that best describes the measured sample spectrum by minimizing a functional that defines the deviation between the spectra.
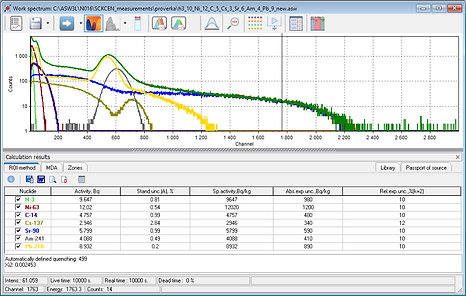
Spectrum of a sample containing H-3, Ni-63, C-14, Sr-90, Cs-137, Am-241, Pb-210 on a logarithmic scale processed by ASW3L software.
The model spectrum is shown in green.

Spectrum of a sample containing Pb-210 and Ra-226 processed by SpectraDec software.
The combined standard uncertainty of the activity of the j-th radionuclide in the measured sample is determined by contributions from counting statistics, the quality of spectral fitting, and the uncertainty of detection efficiency. The latter includes contributions from the counting statistics of calibration measurements, certification of the activity of the reference solution, the mass of the reference solution used in the calibration sample, and the uncertainty of the quenching parameter.
Therefore, the accuracy and reliability of the described method are mainly determined by the quality of the reference solutions used for preparation of calibration samples, the chemical and radionuclide composition of the measured sample, and the counting statistics (measurement time).
In practical terms, the described approach enables identification of alpha- and beta-emitting radionuclides, analysis of complex mixtures without prior separation, rapid screening of water and liquid samples, and reduction of radiochemical procedures in many cases.
Implementation of the method
The liquid scintillation spectrometry algorithms described above for decomposition of liquid scintillation spectra into individual components (radionuclide identification), as well as for calculation of their activity, uncertainties, and detection limits (MDA, decision threshold, detection limit) within the optimized spectral region, are implemented in the SpectraDec and ASW3L software packages.
In practice, ASW3L is typically used for routine measurements and for analysis of relatively simple mixtures (up to approximately 10 components), whereas SpectraDec is applied for detailed spectral decomposition of complex radionuclide mixtures. This combination allows flexible selection of the analysis approach depending on the complexity of the sample and the required level of detail.
In addition to the TRIEL spectrometer, SpectraDec is also used for liquid scintillation analysis and mathematical processing of spectra obtained with other liquid scintillation counters, including Tri-Carb, Quantulus, and similar instruments.
During long-term and intensive application of the described method, rapid LS analysis has been used for measurement of environmental and technological samples of various origins.
Based on extensive testing and practical experience [1], LS screening of natural water samples shows that exceedance of control levels for alpha emitters is mainly associated with the presence of Ra-226 and its daughters.
Thus, the LS screening method makes it possible to rapidly determine the composition of the main alpha- and beta-emitting radionuclides or, at a minimum, to define whether their activities exceed the corresponding control levels.
In a number of cases, this makes it possible to avoid time-consuming radiochemical procedures that would otherwise be required in a conventional approach without prior information about the sample composition.
